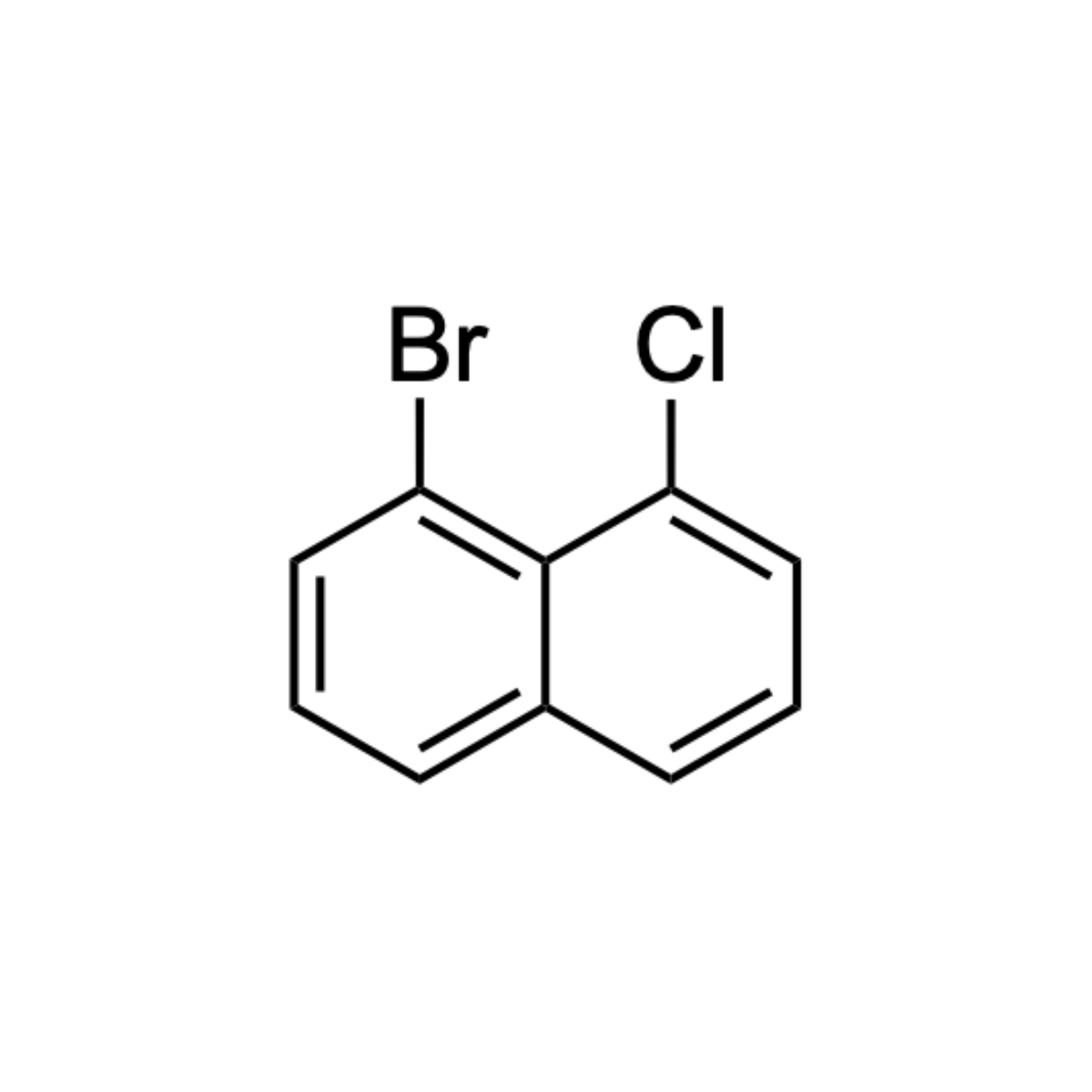
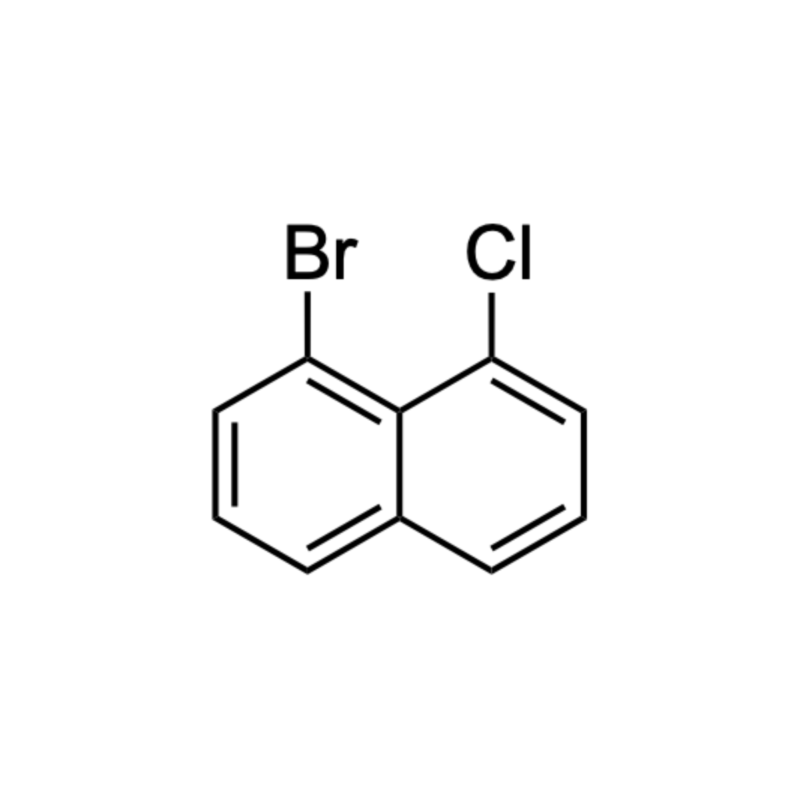
1-bromo-8-chloronaphthalene
IUPAC Name: 1-bromo-8-chloronaphthalene
Synonyms: Naphthalene, 1-bromo-8-chloro-
Catalog NO.:
CAS NO.: 20816-79-9
Brand: Daisa Biopharma
Category: Intermediate, Naphthalene
Tag: 4-1-Bromo-8-chloronaphthalene, Adgrasib intermediate
Molecular Formula: C10H6BrCl
Molecular Weight: 241.51
Pack Size Available: 25 g, 100 g, 1.0 kg
Place of Origin: India
Product Description
Due to its polycyclic aromatic structure, the compound exhibits typical aromatic stability and is poorly soluble in water but readily dissolves in common organic solvents such as ethanol and diethyl ether. The presence of bromine and chlorine atoms enhances its chemical reactivity, making it a useful intermediate in organic synthesis, particularly in the preparation of pharmaceuticals and agrochemical compounds.
The molecule undergoes electrophilic aromatic substitution reactions, and its halogen substituents can also act as leaving groups in various nucleophilic substitution or cross-coupling reactions. As with many halogenated aromatic compounds, appropriate safety measures should be followed during handling, as exposure may be harmful through inhalation or ingestion.
Appearance: White to Brown powder to crystal
HPLC purity: ≥98% a/a
NMR: Confirm the structure
Physical Form: Solid
Melting Point: 87-88 oC
Shipping Temperature: Keep the container tightly closed in a dry, well-ventilated area. Containers that are opened must be carefully resealed and kept upright to prevent leakage. Inert atmosphere, Room Temperature
Storage Temperature: Keep container tightly closed in a dry and well-ventilated area. Containers that are opened must be carefully resealed and kept upright to prevent leakage. Inert atmosphere, Room Temperature (Recommended in a cool and dark place, <15°C)
Inchi Code: 1S/C10H6BrCl/c11-8-5-1-3-7-4-2-6-9(12)10(7)8/h1-6H
Inchi Key: JHZQEADUKRNQBX-UHFFFAOYSA-N
SMILES: ClC1=CC=CC2=CC=CC(Br)=C12
Recommended Products & New Products
About Us
We are a multidisciplinary research organization specializing in Medicinal Chemistry, Process Chemistry, and Biocatalysis—delivering scalable, efficient, and sustainable solutions for complex scientific challenges.
Our medicinal chemistry division drives early-stage drug discovery through the design and synthesis of novel chemical entities, SAR exploration, and creation of natural-product-inspired molecular libraries.
We develop safe, robust, and scalable synthetic routes for APIs and intermediates. Our expertise covers QbD-driven development, impurity profiling, polymorph optimization, and complete CMC/DMF support.
Our biocatalysis services include enzyme screening, whole-cell bioconversion, and industrial bioprocess scale-up—enabling greener, more cost-effective, and sustainable alternatives to traditional chemical transformations.